Have you ever experienced when you rub the comb on pull over, you can pick the small pieces of paper or when you rub the balloon, and it will stick to yourself. Another powerful example is the thunderbolt of lightning during heavy rainy season.
These all are the examples of static electricity.

What is Static Electricity..?
So what is static electricity actually..? The static electricity is basically the imbalance of charge produced by mechanical movement between two bodies’ surfaces.
One of the body is the bad conductor of charge called insulator when rubbed against a material surface it causes the resistance/friction that in turns creates “static charge”.
Science behind Static Electricity:
Actually all the matter exist in the universe is made of tiny particles called “atoms”. These atoms are further broken into 3 basic constituents called “electrons”, “protons” and “neutrons”. The matter is classified into elements in periodic table. There are 118 elements in periodic table till now. The atom is electrically neutral because of equal number of protons and electrons. The electrons are loosely bonded and can escape away the shell of atom upon small excitation energy or mechanical movement like friction.
The neutrons and protons are tightly bonded together in the nucleus of atom. The nucleus is the heaviest part of atom. The number of protons forms the identity of element. It is near impossible to extract / kick off proton from nucleus. Because if we can do this we could have changed the nature/identity of element. However neutron can be kicked off nucleus and as a side effect emits radioactive waves. Now when the electron from the valence shell is removed or electron added into the valence shell then it will become positive charged or negative charged respectively.
Benjamin Franklin’s Experiments Observations:
The fluid model of static electricity, was discovered by early scientist and pioneer researcher named Benjamin Franklin. He witnessed that upon rubbing glass rod with silk cloth will cause force of attraction between the two.
When the wax was rubbed against wool cloth this will also cause force of attraction between the two.
It was also observed that if two glass rods were rubbed with their respective silk cloths then these two glass rods repel each other. Hence generating force of repulsion.
Another observation was that when glass interacted/rubbed with silk and wax interacted/rubbed with wool then wax and glass would attract one another.
Hence it is was speculated by Franklin that some sort of invisible “Fluid” is transferred between two bodies during the process of rubbing. This transfer of fluid would render one body positively charged and another negatively charged. This positively charged and negatively charged were related to the deficiency and excess of that “fluid”. This hypothetical transfer of fluid then become “Charge”.
Hence it is was postulated that charge that is created by rubbing wax was negative and that charge created by rubbing wool is positive.
Charles Coulomb Measurement:
Charles Coulomb used the special device called “Torsional Balance” to measure exact value of charge. His experiment than came to following result
“If two point objects equally charged to 1 coulomb having no physical mass are placed at a distance of 1 meter apart, then there exist a force of 9 billion Newton either attractive force or repulsive force for opposite charges and similar charge types respectively. ”
The unit of measurement of charge was dedicated to the name of Scientist “Coulomb”.
1 Coulomb of charge is actually the excess or deficiency of electrons. Or conversely speaking 1 electron charge is equal to Coulomb (C)
Where F = Electrostatic Force
k = Coulomb’s Constant =
q1 and q2 are two point charges
r = distance between two point charges in meters
Static Electricity Phenomenon In terms of Electronic Charge:
When the two neutral bodies/materials are brought close together and rubbed with each other, this will create movement in electrons. The electrons will start to leave from one body and enter other body. The body that releases electrons is said to be positively charged due to scarcity of electrons and the body that receives electrons is said to be negatively charged due to excess of electrons.
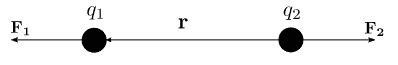
Examples of Static Electricity:
Hat and Hair Example of Static Electricity:
In the context of all discussion above it is now clear that when we take off the hat our hairs stick to the hat because of transfer of charges/electrons from hair to hat. This will create negative (excess of electrons) static electricity on hat and positive (deficiency of electrons) static electricity on hair.
Static Electricity Balloon Example:
We can also say that a charged object will attract neutral object because of the same electrons flow from charged object to neutral. Example of this is a balloon that when rubbed on your hair will get negative charge, then it is brought near to the neutral wall but the balloon will stick to the wall because of electron flow from balloon to wall. This is also true for the case when we brush our hair with comb then the comb can pick up small pieces of paper.

Static Electricity of Clouds Friction:
The friction in the clouds in rainy season cause the generation of static electricity. This static electricity is stored in the clouds but is visible due to millions of volts created spark in sky. This static electricity converts into electrical current when some sort of current path is generated from clouds to the ground like a kite can bring the thunderbolt to the earth surface.

Ozone Cracking:
The ozone is created due to static discharge. This ozone is not good for elastomers. This ozone can make deep cracks in vehicle components like O-rings. The damaged fuel line from ozone can cause fire. To protect from this use elastomers that resist ozone.

Static Electricity vs Current:
The charged objects will hold these states of excess / scarce electrons until it is applied by external force to move it in a particular direction. These electromotive force (EMF) or “voltage applied across” will cause the electron to flow thus converting static electricity into “current”.
Current is always moving in a direction through a metallic wire. While static electricity remain stored in a body when applied to mechanical friction/movement.

